Allergic to peanuts? New protocol being tested at USF may make your life easier
Living with a peanut allergy can be challenging, but a peanut protein being tested at the University of South Florida may help children and adults increase their tolerance.
When you’re allergic to peanuts, even a tiny portion of a peanut may cause a reaction. Yet avoiding them may be difficult: The person next to you in the lunchroom may be eating a peanut butter and jelly sandwich, or your almond butter may be contaminated with peanut dust at the packinghouse.
When Harry Coleman III was traveling in Mexico, he and his family had complications because the label was written in a foreign language.
“We had checked it [a candy label],” the South Tampa man recalls, “but did not realize that there were many variations of the word in Mexico.”

His son, Harry Coleman IV, who goes by Will, then ate a piece of chocolate candy that Coleman believed was OK for him. Not so.
Will already had been hospitalized at age 1. He’d only taken one bite of a tortilla smeared with peanut butter, and spit it out. It wasn’t fast enough.
Research reported by the American College of Allergy, Asthma and Immunology suggests close to 2.5 percent of children in the United States may be allergic to peanuts. It also suggests the number of children with peanut allergies has increased 21 percent since 2010.
Peanut allergy sufferers are less likely to grow out of it than those with other allergies, which makes getting a handle on it early more important.
Yet avoidance and, when that fails, antihistamines or epinephrine, have been relied-upon treatments. Soon there may a better way: Will is among about 500 nationwide who participated in the Phase 3 trial of a peanut protein to help desensitize people to the legume.
Some 24 have participated in a series of tests on the protein, known as AR101, at the University of South Florida.
Among those who completed the trial, 96.3 percent tolerated 300 milligrams of peanut, compared to 8.6 percent taking a placebo, according to the study’s sponsor, the Brisbane CA-based Aimmune Therapeutics Inc. The numbers decline when the dose is increased – to 84.5 percent at 600 milligrams and 63.2 percent at 1,000 milligrams. Those taking the placebo were logged at 4.3 percent at 600 milligrams and 2.6 percent at 1,000 milligrams.
One peanut kernel contains 300 milligrams of peanut protein.
“Volunteers for studies like this are heroes — without them, we can’t push the window,” says Dr. Richard Lockey, Professor of Medicine, Pediatrics and Public Health Director, Division of Allergy/Immunology, at USF.
Identifying the problem
Dr. Lockey says a peanut allergy can be diagnosed by a patient’s history, rather than a test. The test can confirm it and, to some extent, determine the degree of sensitivity.
Only about 10 percent of the people suffering adverse effects from a food is allergic. The other 90 percent are experiencing side effects, like flatulence after eating beans or belching after consuming onions, he points out.
Peanuts are popular because they are a great source of protein and essential fats. Because they are cheap, they can be considered an ideal food. Yet for those who are allergic, they can be dangerous or even deadly.
An estimated 4 to 6 percent of children in the United States have food allergies, according to the Centers for Disease Control and Prevention. Eight foods, including milk, eggs, fish, crustacean shellfish, wheat, soy, peanuts, and tree nuts, cause 90 percent of the serious reactions.
When an allergy exists, the body’s immune system mistakenly identifies a food as harmful, causing a “specific and reproducible immune response,” it says.
An allergy can cause any of a number of symptoms including vomiting, abdominal upset, hives, itching, runny nose, swelling, wheezing, diarrhea, and anaphylaxis, a life-threatening reaction that can involve swelling in the throat and constricted airways in the lungs.
A 2015 study by the Johns Hopkins Bloomberg School of Public Health, reported in Science Daily News suggests there is a genetic link involved in food allergies.
In Will’s case, no one in his household suffers from peanut allergies. It wasn’t until after he began reacting that the family discovered there was one family member with a mild allergy on his mom Debra’s side of the family.
Reducing sensitivities
New peanut allergy guidelines, from a panel sponsored by the National Institute of Allergy and Infectious Diseases, suggests peanuts be introduced into infants’ diets early — perhaps at 4 to 6 months of age — to reduce the likelihood of allergy.
Parents and caregivers are advised to check with their healthcare providers beforehand. A blood or skin test or oral food challenge may be used to decide now to introduce peanuts safely, according to the National Institutes of Health, the nation’s medical research agency.
Regular consumption of peanuts beginning in infancy, and continuing until the age of 5, has been shown to reduce peanut allergies by 81 percent in infants considered high risk because of severe eczema, egg allergy or both.
The Learning Early About Peanut Allergy study was funded by NIAID.
Introducing infants to all foods by eight months of age, or no later than one year of age, appears to create tolerance rather than allergies, Dr. Lockey says.
The peanut study
Will is pretty much a typical patient in the peanut studies at USF. Patients are seen at USF’s Asthma, Allergy and Immunology Clinical Research Unit, located in a building visible from Florida Hospital’s Emergency Room at the northern edge of USF’s Tampa campus.

“He’s the youngest in our study. We’ve had two that were 5 when they started,” says Michelle Twitmyer, a Clinical Research Coordinator for USF Health.
Parents seem more motivated than those who continue to have peanut allergies into adulthood.
“The majority of patients have been kids, even when they allowed adults,” says Dr. Thomas Casale, the principal investigator and a professor in Internal Medicine at USF’s Morsani College of Medicine. “I think it’s a bigger problem with kids.”
“The parents are highly motivated to keep their children alive,” Twitmyer explains.
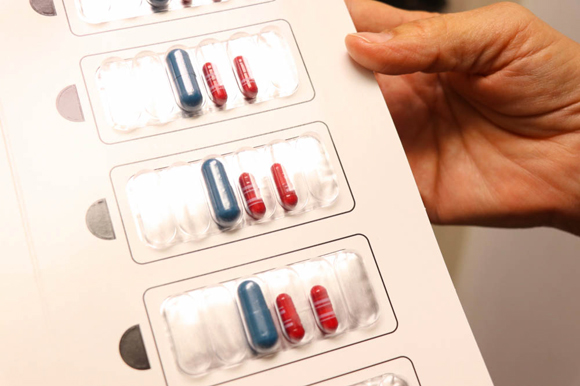
The protocol begins with two days of intensive testing with increasing doses, after which patients go home with a dose equivalent to 1/100th of a peanut.
Will started taking part in the study about a year ago, right when he turned 4, although he was initially given a placebo. He was switched to the protein powder Feb. 22, 2018, Twitmyer says.
At a recent visit, Will kept himself entertained playing Ramses Pyramid on a twin bed at the unit.
He says he likes the fact that these doctor’s visits might help him. He also indicates he likes peanuts and wishes he could eat them like his friends.
“I don’t want to be allergic at school,” says the 5-year-old, who has begun kindergarten at Mitchell Elementary.
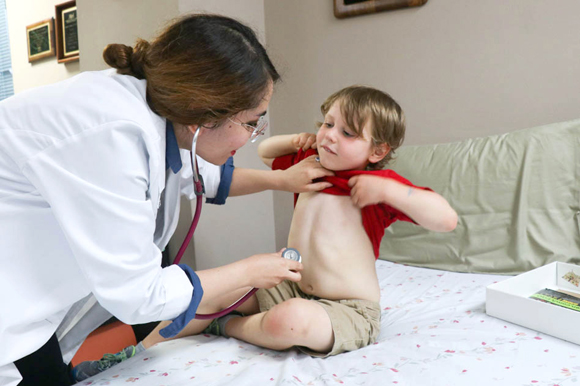
So, like he does every two weeks, Will is examined. Based on his experiences since the last visit, he will be maintaining his dose of 120 milligrams of AR101, which is mixed into a food like applesauce or pudding.
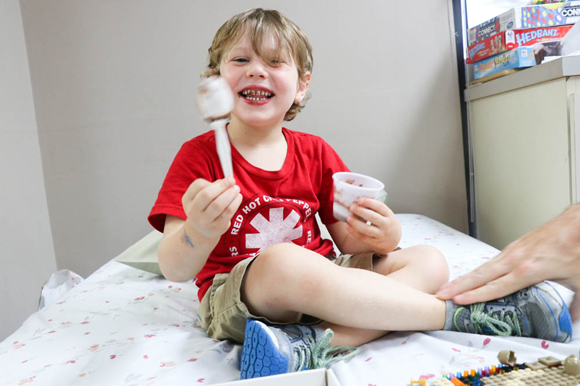
This time Will chooses chocolate pudding. After he eats it, he hangs out to see if he will develop a reaction like itchiness or hives. He waits for half an hour, because the dose is the same, and has no issues.
On days that his dose increases, he stays for an hour and a half. When he’s in the clear, he leaves for home with a kit containing capsules he can take daily.
“It definitely feels like we’re going in the right direction,” his dad says.
While increasing tolerance may not eliminate the allergy, it can significantly reduce problems that result when foods are contaminated by peanuts.
“The amount that they could tolerate is typically important in accidental exposures,” Dr. Casale says. “That’s what’s really been the problem.”
A better future
Aimmune Therapeutics plans to apply for a Biologics License Application with the FDA by the end of the year for AR101, followed by a Marketing Authorisation Application from the European Medicines Agency in 2019.
“In the United States, AR101 has FDA Fast Track Designation, as well as FDA Breakthrough Therapy Designation for peanut-allergic patients ages 4 to 17, so the timeline to potential approval could see it become available later in 2019,” says Alison Marquiss, the company’s VP for Community and Public Affairs.
“For me it’s encouraging knowing what we are doing here is really changing what’s happening in the real world,” says Catherine Renee Smith, a Head Coordinator and Clinical Research Associate at the Research Unit. “It can change lives. Once the FDA gives the approval, it’s going to be widely available to everybody in the U.S.”
In the meantime, young children with peanut allergies still can choose to participate in a study. Enrollment is expected to open next spring for 1 to 3-year-olds, Dr. Lockey says.
If you’d like to participate in the study, please call 813-631-4024. To have a child assessed, call 813-971-9743 for a doctor’s appointment and evaluation.
“You have to have a bona fide reaction to peanuts,” Dr. Lockey explains. “It’s not hard [to get in] if you’re really peanut allergic.”
Want to make a donation to support USF research? Visit the USF Foundation website and click on Where to Give in the dropdown menu.