TGH begins trial of new device created to destroy tumors without surgery
Tampa General Hospital begins testing a new procedure using sound waves to remove tumors without surgery, a non-invasive method that could lead to quicker recoveries and FDA approval for more widespread use.
Tampa General is among seven U.S. healthcare providers selected to take part in a groundbreaking study on using ultrasound waves in non-invasive surgeries to remove tumors.
Physicians at the hospital conducted the first use of a novel device called HistoSonics System in January using a new technique called Histotripsy. This method is very much in its investigative stages and offers hope for success at other centers taking part in the trial to gain FDA approval.
HistoSonics has been working on developing this new technique in partnership with the University of Michigan. A patient who has liver tumors that have been deemed inoperable can qualify to take part in the #HOPE4LIVER trial.
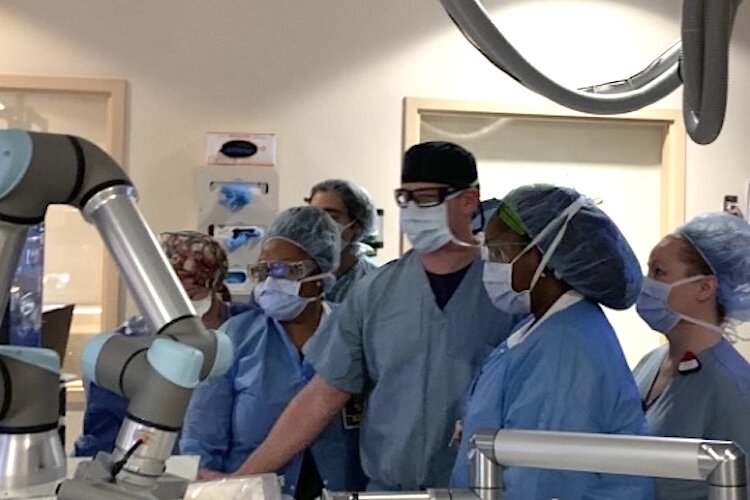
While conducting this procedure, the patient will be put under general anesthesia, not because of pain, but to keep the patient from moving. Meanwhile, the Histotripsy device uses its robotic arm to perform the ablation of the tumor using ultrasound waves.
“These are normal ultrasound waves, but they are set to a specific frequency that causes air bubbles to form in cells,” says Dr. Cliff Davis, assistant professor and Director of the Interventional Radiology Residency Program at the USF Health Morsani College of Medicine and principal investigator of the #HOPE4LIVER trial at the TGH. “This then causes the cells to die immediately.”
The procedure enables physicians to target a specific area precisely within 1/16th of an inch causing these air bubbles to form and rupture the cells without using any type of incision or probes. The debris is then removed.
“Everything is done like a normal ultrasound outside the body,” Dr. Davis says. This should kill the cells immediately.
An MRI performed immediately after the first Histotripsy procedure at TGH showed a perfect hole where the tumor had been. The procedure was deemed a success. A month later, the hole remained as expected with no new tumor. This procedure can take anywhere from 20 to 45 minutes based on the size of the lesion.
“As far as the plan, this device is not FDA approved yet and that is what this trial is intended to do,” Dr. Davis says, “It’s to demonstrate safety and efficacy, that’s called a Phase 1/ Phase 2 trial. The intention is to prove that if we target a tumor and we ablate it, that it is successful. And that we get all of the tumor that we are targeting.”
Tampa General was selected for the #HOPE4LIVER trial because it treats a large number of liver cancer patients and has been a major liver transplant center since 1987, now performing about 100 per year.
“I was really excited about this technology when I heard about it because I truly believe it’s going to change medicine and at some point, it’s going to completely change the way we treat tumors and even perform surgery in the future,” Dr. Davis says. “The ability to kill cells with a target without cutting the patient open or using heat or cold is really amazing because we feel that that will be much safer.”
Histotripsy results from animal trials, which have been going on for about 10 years, have shown that this technology is very safe. The hope is that in the progression of this technology, this method of procedure will be available to use throughout any organs in the body to treat patients.
“This is the surgery of the future; to be able to cut or destroy tissue without cutting the skin open, without bleeding, and hopefully with less pain and quicker recovery,” Dr. Davis says. The goal, he says, is to find more patients who match the criteria to undergo this treatment, potentially enabling several more procedures within the year and leading to FDA approval.
The HistoSonics trial is one among many taking place at Tampa General.
“Tampa General is pioneering new ways to save lives by testing innovative solutions to the most difficult medical problems,” says Tampa General President and CEO John Couris in a news release. “As an academic medical center, discovering new treatments through this kind of groundbreaking research is a vital part of our mission to deliver world-class care to our patients.”
For more information, visit:
- Interventional Radiology Residency Program at USF Health
- Tampa General Hospital
- HistoSonics
- Dr. Cliff Davis